Ready.
Aim.
Fire.
And just like that, a drone, carrying life-saving medical supplies, cuts what was once a two-hour journey over rough terrain down to 15 minutes.
Tops.
And in so doing, saves countless lives, reduces waste around spoilage of blood and medicines, and means fewer trucks on roads already susceptible to highway robbery, hijackings, or flooding.
Sounds like science fiction, right? Well, it’s not and it’s happening in parts of Rwanda as we speak.
But what if you do not operate in the far-flung reaches of the Middle East, Central Asia, or Africa? Or (and we’ll do you one better here): What if you do not have the funding for this sort of cutting-edge technology?
No matter, as cold-chain technology, or those temperature-controlled logistics products necessary in the delivery of pharmaceuticals to end-users the world over, are looking to take off (no pun intended!) with supply unlikely to meet demand until 2022, at the soonest.
This is not to say technology should be dismissed when optimizing the cold chain; rather, accurate tracking software and monitoring devices are absolutely imperative in the movement of pharmaceutical products since tracking package integrity means no risk of spoilage or worse, legal action.
Current Operational Environment:
As stated, the demand for cold-chain technology looks to remain strong through the early part of the next decade and will bring with it various opportunities and threats.
Regarding the former, the market looks to grow 56% to $15B with pharmaceuticals, biotech, vaccines, blood products, and clinical materials accounting for almost 13% of that increase.
And while the sector looks promising from a profits-based perspective, there are potential pitfalls that could easily result in big losses for the would-be cold shipper who fails to perform his or her market research.
Consider, and despite improvements in freezer technology, that cold chains consume 20% more fuel than more traditional supply chains. Coupled with the increasing standards around sustainability as put forth by national and international governing bodies and the logistician looking to break into this field would be wise to invest in optimisation software as well as tracking technology to ensure no break in the integrity of the entire pipeline.
Product requirements for maximum social effect:
Sourcing such software and monitoring devices for pharmaceutical cold chaining should not be dismissed or left until the last minute.
Investing time upfront will not only ensure quality standards are met and maintained from port to patient, but also reduce the amount a shipper has to spend on making more robust his or her packaging.
Placing a temperature, tilt, and impact enabled sensor on a package is a much easier and proactive approach in managing medical shipments while simultaneously enabling the supply chain manager to learn which routes are best to use based on real-time feedback.
This end-to-end, network-based approach to meeting a social need means a cold-chain specialist not only maximises the likelihood of delivering much-needed medical supplies to those in crisis, but also reduces regulatory issues by avoiding those ports unwilling to comply with Good Distribution Practice (GDP) guidelines.
Making warm the cold chain:
If not made clear already, there is a distinct competitive advantage up for grabs by those supply chain managers looking to enter the pharmaceutical shipment arena.
Many companies, especially those conducting clinical trials, are now no longer able to keep in-house their own confidential shipping procedures on account of the wave of new products that necessitate the movement of human cells, genetically-modified products, and other sensitive information.
In endeavouring to embark on fulfilling this need, the cold-chain logistician should source technology and software with an eye on both the $3.7B market as well as security as a breach in data could mean a loss of potentially life-saving research.
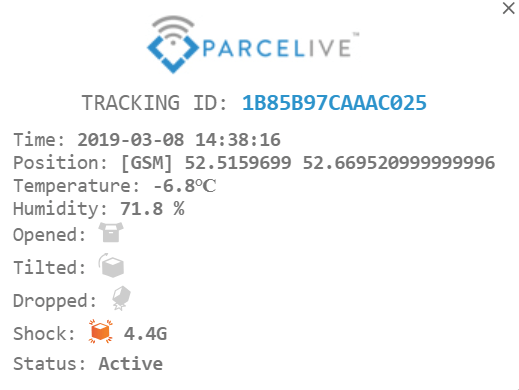
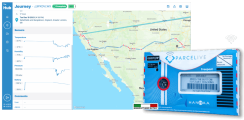
Enter ParceLive, and while it is not as ‘cool’ (again, no pun intended) as a drone, the real-time alerts and audit updates alert the shipper to exceptional events so as to ensure that no pharmaceutical product is dropped, mishandled, left out in the sun, or otherwise tampered with.
What’s more, the technology takes the virtual tracking to the physical world with recyclable GPS transponders, which marries nicely with the greening-aspect of the cold chain.
In saving money, these same transponders work for bulk shipments of medical supplies (think disaster relief efforts) just as they would for small batch orders (here, clinical trials).
Ultimately, and unlike an unidentified flying object over Rwanda, ParceLive is scalable with no known geographic limitations and, therefore, no barrier to the amount of environmental, social, and human good a cold-shipper can do when looking to move pharmaceuticals the world over.